|
8/31/2023 0 Comments Group 7 reactivity trendThe process is assumed to start from the solid sodium halide. The table below shows the energy changes that vary from halogen to halogen. The total enthalpy change is the sum of the enthalpy changes for the halide ion half-reaction and the sulfuric acid half-reaction. That is the same irrespective of the halogen in question. Heat is emitted when the changes involving the sulfuric acid occur. This is not the total enthalpy change for the whole reaction. The diagram shows that the overall change involving the halide ions is endothermic (the green arrow is pointing up toward a higher energy). The enthalpy change shown by the green arrow in the diagram for each of the halogens must be compared. The figure below shows how this information fits together: The value of interest is the reverse of atomization energy.Ītomization energy is the energy needed to produce 1 mole of isolated gaseous atoms starting from an element in its standard state (gas for chlorine, and liquid for bromine, for example - both of them as X 2). To account for this, it is simpler to think in terms of atomization energy rather than bond energy. In bromine and iodine, heat is also released during condensation to a liquid or a solid, respectively. Energy is released when these bonds are formed. The energy recovered when the chlorine atoms convert to diatomic chlorine.This is the reverse of the electron affinity of the chlorine (the electron affinity can be acquired from a data table and negated). The energy required to remove the electron from the chloride ion.The energy required to break the attractions between the ions in the sodium chloride (the lattice enthalpy).Taking sodium chloride as an example, the following energetic quantities are important: The amount of heat evolved or absorbed when a solid halide (like sodium chloride) is converted into an elemental halogen must be considered. Hydrogen sulfide gas can be detected by its "rotten egg" smell, but this gas is intensely poisonous.Įnthalpy change variation between halogens The red color is due to the I 3 - ion formed by reaction between I 2 molecules and I - ions. There is also a red color where the iodine comes into contact with solid iodide salts. The reaction is exothermic: purple iodine vapor is formed, with dark gray solid iodine condensing around the top of the reaction vessel. This is confirmed by a trace of steamy fumes of hydrogen iodide and a large amount of iodine. The half-equation for its formation is as follows:Ĭombining these two half-equations gives the following net ionic equation: 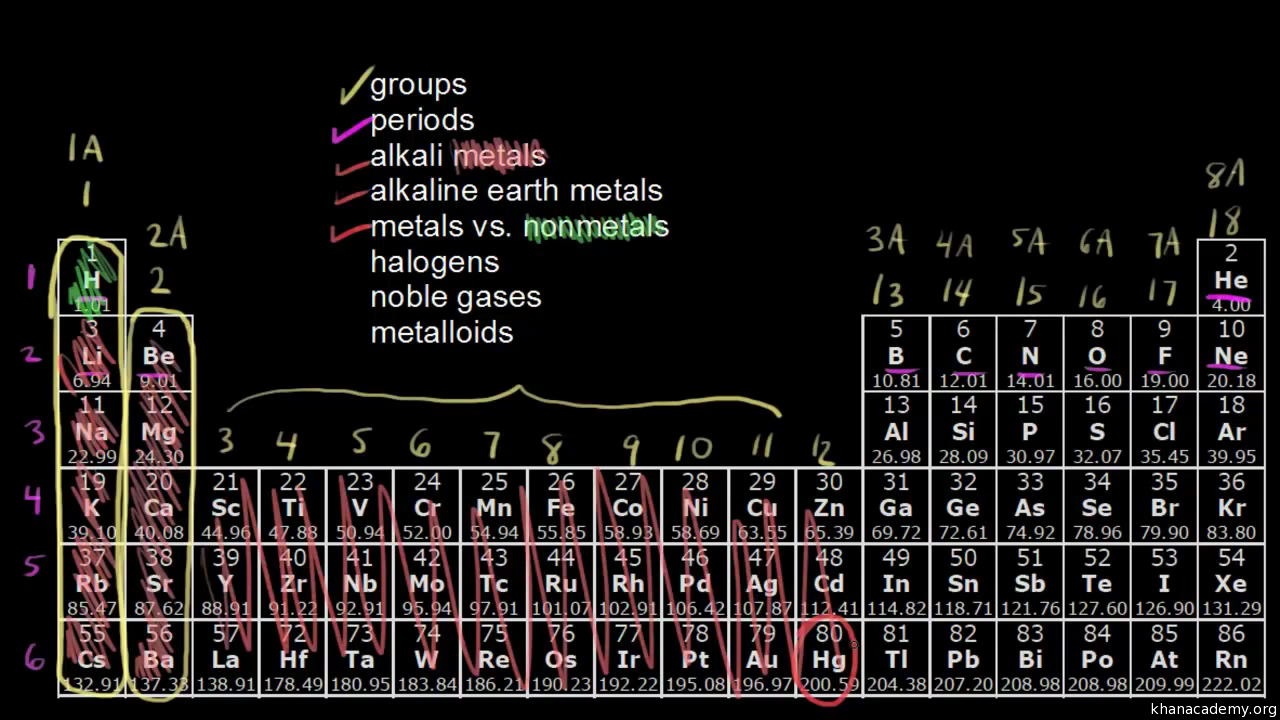
The most abundant product is hydrogen sulfide. sulfur to hydrogen sulfide (sulfur oxidation state = -2).sulfur dioxide to elemental sulfur (oxidation state = 0). 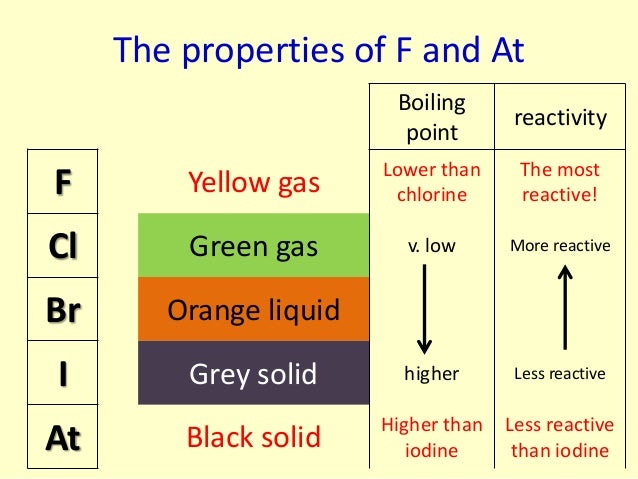 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
